Proyecto Integrado
lunes, 17 de noviembre de 2014
Objective
The objective of our experiment is to proof if there is any kind of relationship between the height of the resulting black-foam structure and the amount of p-nitro aniline (C6H6N2O2) that reacts with a constant amount of hydrosulfuric acid (H2SO4)
Hypothesis
Whenever the amount of solute changes we will appreciate a direct relationship between the volume of the resulting foam structure and the amount of solute (p-nitroaniline) in the solution (p-nitroaniline and hydrosulfuric acid).
Variables
- Dependent variable: The resulting foam structure
- Independent variable: The amount of p-nitroaniline for each trial
- Controlled variable: Amount of sulphuric acid (5ml), the container in which the reaction takes place and the concentration of the hydrosulfuric acid.
Materials
To work out the molarity of the sulphuric acid knowing that the concentration is 96% and that the density in one litre is of 1840g, we work out the mass and divide it by the molecular mass.
96% of 1840= 1766,4g
Molarity= 1766,4/ 98,07= 18,011 g/mol
96% of 1840= 1766,4g
Molarity= 1766,4/ 98,07= 18,011 g/mol
- P-nitroaniline
- Spoon
- Scale
- Bunsen burner
- Tripod for the bunsen burner
- Pipette
- Goggles
- Masks
- Flask
- Small bowl (container)
miércoles, 8 de octubre de 2014
Method of the experiment
- Measure the amount in grams of p-nitro aniline for that particular case: we will be making 5 trials by each measurement, in order to obtain reliable data. These are the seven amounts of p-nitro aniline (g): 5; 6; 7; 8; 9; 10; 11.
- Connect the bunsen burner until a visible (between blue and orange colour) flame appears.
- After having measured the amount of p-nitro aniline, we will place the small bowl on top of the bunsen burner and spill 5 ml of hydrosulfuric acid H2SO4.
- Wait until it heats up and the reaction takes place.
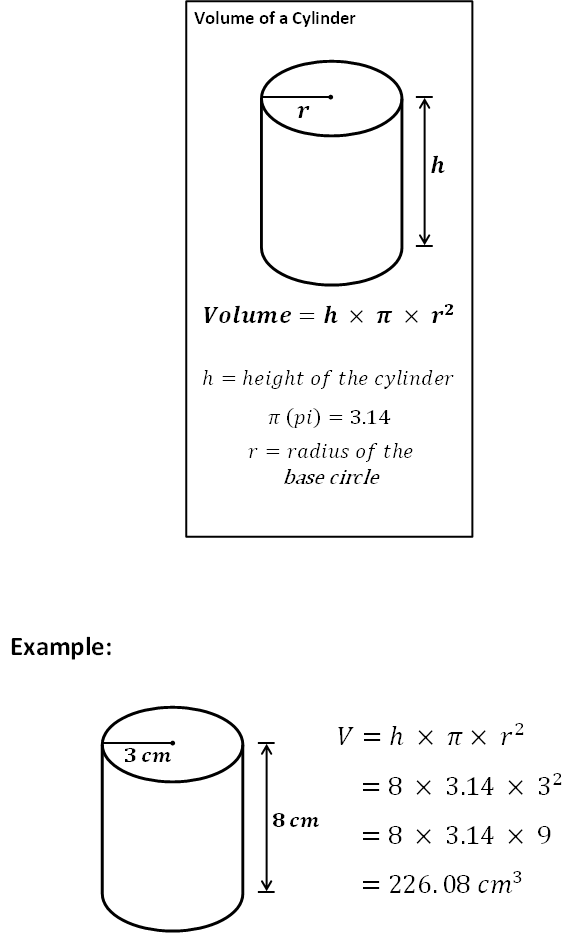
- After exploding we will measure the height of the black-foam structure from one of its sides, so that if it is tilted we won't take into account the difference in the curve. We will ass well measure the diameter of the black-foam structure (for this take a bottom measurement of the structure and one from the top of it and make an avarage), that will be very similar to a cylinder.
- Make a table with the collected data.
- Calculate the aproximate volume for each black-foam structure, through this formula:
- Finally observe if there is a direct relationship between the amount of p-nitro aniline and the aproximate volume of the black-foam structure, that will increase every time more p-nitro aniline reacts with the hydrosulfuric acid.
miércoles, 24 de septiembre de 2014
Bibliography
- Es.wikipedia.org,. (2014). P-nitroanilina. Retrieved 17
November 2014, from http://es.wikipedia.org/wiki/P-nitroanilina
- Wikihow.com,. (2014). Volume of a
Cylinder Cheat Sheet - wikiHow. Retrieved 17 November 2014, from
http://www.wikihow.com/Sample/Volume-of-a-Cylinder-Diagram
- YouTube,. (2014). EXP 1.
Explosive Polymerization of p Nitro Aniline. Retrieved 17 November 2014,
from https://www.youtube.com/watch?v=h4pNXAtPJp8
- Poshkus, A., & Parker, J. (1970). Studies on nitroaniline–sulfuric
acid compositions: Aphrogenic pyrostats. Journal Of Applied Polymer Science, 14(8),
2049-2064. doi:10.1002/app.1970.070140813
Es.wikipedia.org,. (2014). P-nitroanilina.
Retrieved 17 November 2014, from http://es.wikipedia.org/wiki/P-nitroanilina
Wikihow.com,. (2014). Volume of a Cylinder
Cheat Sheet - wikiHow. Retrieved 17 November 2014, from http://www.wikihow.com/Sample/Volume-of-a-Cylinder-Diagram
YouTube,. (2014). EXP 1. Explosive
Polymerization of p Nitro Aniline. Retrieved 17 November 2014, from
https://www.youtube.com/watch?v=h4pNXAtPJp8
Poshkus, A., & Parker, J. (1970). Studies
on nitroaniline–sulfuric acid compositions: Aphrogenic pyrostats. Journal Of
Applied Polymer Science, 14(8), 2049-2064. doi:10.1002/app.1970.070140813
Poshkus, A. and Parker, J. (1970). Studies on
nitroaniline–sulfuric acid compositions: Aphrogenic pyrostats. Journal of
Applied Polymer Science, 14(8), pp.2049-2064.
Wikihow.com,. (2014). Volume of a Cylinder Cheat Sheet - wikiHow. Retrieved 17 November 2014, from http://www.wikihow.com/Sample/Volume-of-a-Cylinder-Diagram
YouTube,. (2014). EXP 1. Explosive Polymerization of p Nitro Aniline. Retrieved 17 November 2014, from https://www.youtube.com/watch?v=h4pNXAtPJp8
Poshkus, A., & Parker, J. (1970). Studies on nitroaniline–sulfuric acid compositions: Aphrogenic pyrostats. Journal Of Applied Polymer Science, 14(8), 2049-2064. doi:10.1002/app.1970.070140813
Suscribirse a:
Comentarios (Atom)